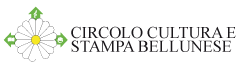5 The “Calchera” (800 m a.s.l.)
How lime was made
Limekilns once played a significant role in local construction and constituted an important source of revenue for the inhabitants of the valley, as the materials involved were easily found on site. The stones necessary for the production of lime were skilfully extracted either from the landslide debris that surrounded the foot of the “Pale” walls or along the Tegnàs riverbed. Bundles of broadleaf branches (beech, hazel, ash and hornbeam) were used as fuel; some of it was leftovers from firewood, or from the charcoal kilns.
 Calchera, or lime kiln, built with dolomite ashlars.
Calchera, or lime kiln, built with dolomite ashlars.
A short history of lime
Lime has been used by humans for millennia. We can never be sure about the exact date of its discovery, but thanks to some finds at human dwellings with lime-plastered and painted in red ochre walls in Mallaha in the Palestine, we can narrow down its first use to approximately 14 to 15 thousand years ago. Widespread use of lime began in Mesopotamia and Anatolia between 7000 and 6000 BC. The Balkan Peninsula saw its arrival around 5600 BC. The process of decarbonation, hydration and re-carbonation became common knowledge more than 2000 years before Christ; the vertical shaft kilns in Mesopotamia date back to that period and they are very similar to the modern ones.
It is accepted that almost all ancient peoples, among which the Egyptians, Chinese, Maya, Phoenicians, Greeks and Romans understood the art of lime burning as well as its manifold use. It was the Romans, however, who brought lime production to perfection and began to use it in a variety of ways. The first detailed and complete written treatise on lime-based mortars happens to be De Architectura by Marc Vitruvius from the 1st century BC.
Lime burning occurs inside structures (kilns or furnaces) that are locally known as calchere. There are various types thereof, with features varying based on the quality of the desired final product.
The initially utilised limekilns were designed with simple features:
 Stack kiln (draw. Francesca Sommavilla)
Stack kiln (draw. Francesca Sommavilla)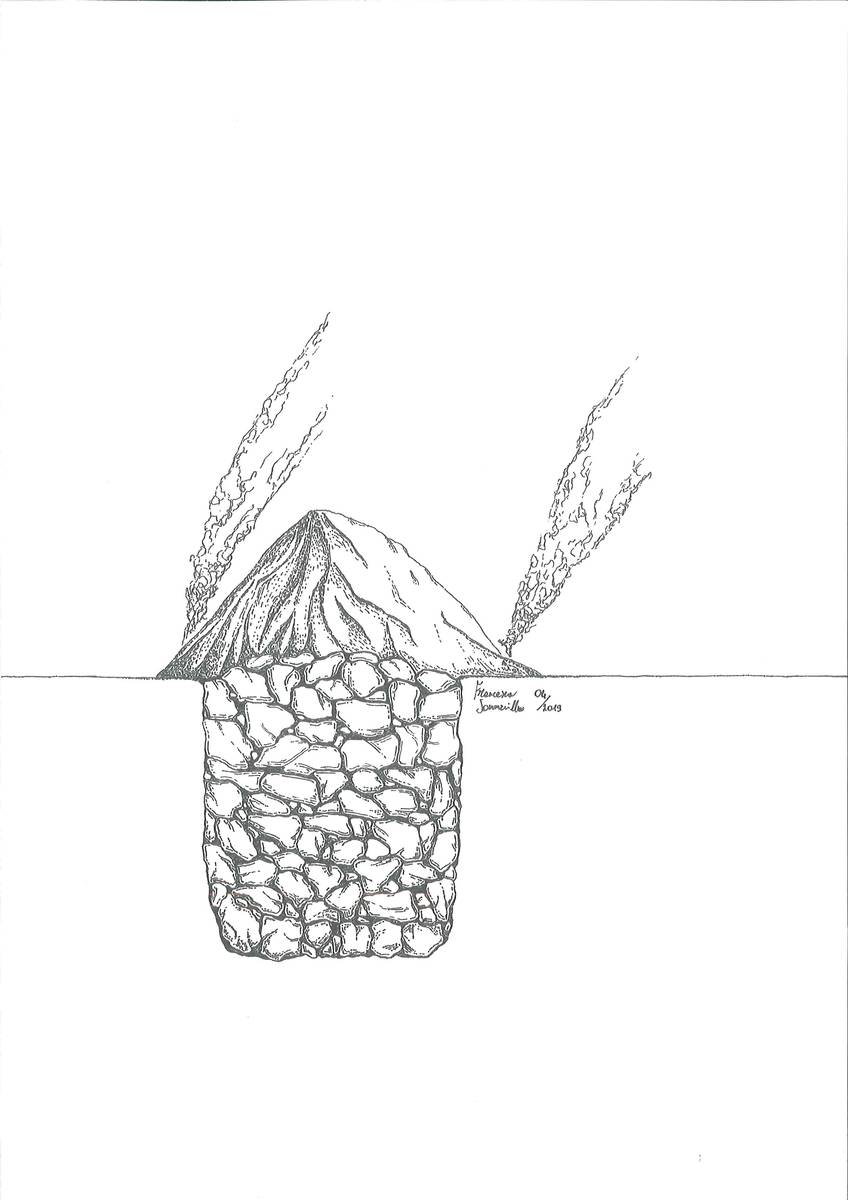 Stationary shaft kilns (draw. Francesca Sommavilla).
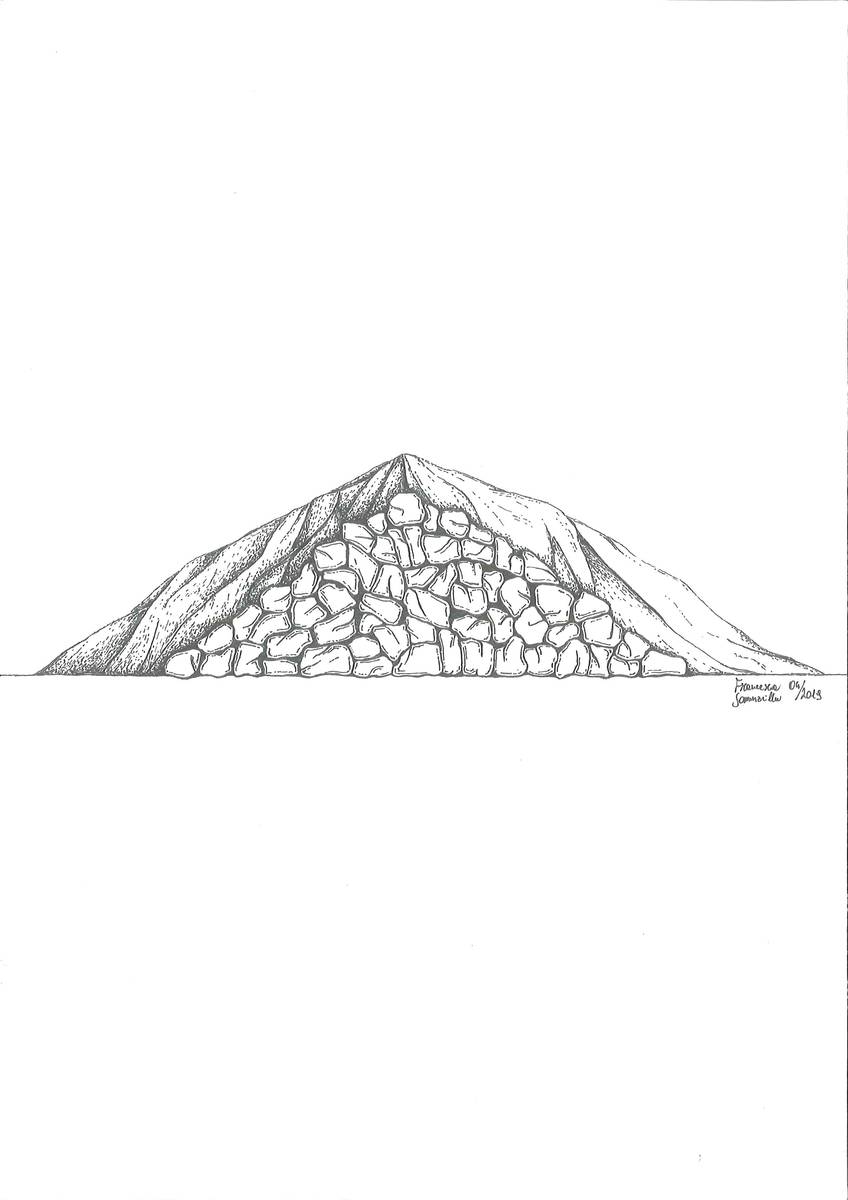
Stationary shaft kilns (draw. Francesca Sommavilla).  Masonry-style "Calchera" (draw. Francesca Sommavilla).
Masonry-style "Calchera" (draw. Francesca Sommavilla).-stack kiln: carbonate stones were piled on a flat surface; the stones were then covered with dry dung and wood gathered from the forest. Once kindled, the fire had to be kept burning until the very end of the lime-burning process.
-stationary shaft kilns: a hole was dug in the ground, choosing clay soil, whenever possible, as it hardened when heated, thus ensuring the thermic resistance; the hole was then filled with carbonate stones and the whole structure was covered with fuel.
As a result, the acquired know-how boosted the technological development that lead to the introduction of built-in kilns, which were still in use in Belluno and its surrounding areas up to several decades ago and can still be found in the Dolomites. The kilns were built on the sides of the slopes in such a way, that, with the exception of their front part, they were completely buried underground. This was done to prevent heat dissipation. Construction consisted of digging a circular hole on the side of slope; it was then completely dry-coated with heat-resistant stones, leaving an opening at the front lower part for fuelling the fire.
The three previously mentioned kilns are intermittent fire kilns. The stones used in these kiln types do not come into direct contact with the fuel, but are merely “licked” by the heat, resulting in obtaining pure-white, high-quality, uniformly calcinated lime, as a final product.
Continuous-fire kilns require the alternation of limestone layers with those of fuel, the resulting product being of doubtlessly much inferior quality, as it was polluted by the fuel residues.
With the arrival of the industrialisation in the nineteenth century, the first great kilns with continuous refilling were introduced. The fuel never came in contact with the processed material, thus obtaining lime free of pollutants.
Construction and use of the “Agordina” limekiln.
In oder to build a “calchera” the terrain had to be cleared, usually in circular shape. The size varied in accordance to the quantity of limestone or dolostone that was intended for processing. The hole was then dry coated with heat-resistant stones. There was an opening for the refilling of the fuel at the lower front part, usually one to one and a half metres large. The kiln’s wall was thicker at the base in order to obtain an indentation of approx. 20-25 centimetres on the inner side of the wall, at a height of around 40 to 50 centimetres from the bottom. This way a small ledge was created on which the blocks of limestone or dolomite were laid in a freestanding dome-shaped heap. The stones were underpinned by a temporary wooden frame in order to facilitate the construction of a vault. These ashlars had to support the rest of the materials thrown in haphazardly into the kiln during the burning process. The stone blocks varied in size and they were chosen in a way to make space for the passage of the heat between them. Once the whole kiln was full, a small canopy was built over it to prevent the fire from extinguishing in the case of rain. The kiln was then kindled using brushwood; the fire had to be kept burning with the help of brushwood or other small branches so that it constantly maintained the necessary temperature of approx. 1000 C° for the decarbonation process. The quantity of the wood necessary for lime production varied according to the size of the kiln. The common ones had a diameter of roughly 3 metres and almost the same height - it is estimated that between 70 and 80 kg of wood were used per 100 kg of ready lime. The production duration itself varied according to the size of the kiln. The above-mentioned variety usually took a week for the burning to be completed.
The produced lime was then put in chests and slaked in order to obtain “grassello”, or lime putty, which as a rule was left to rest and mature in special lime pits for months before it could be put to use.
The chemical process
Aerial lime is obtained by the burning, or calcination, of limestone or dolomite at high temperature between 900 and 1200 °C. By releasing CO2 while burning it at 900 °C, the limestone, or calcium carbonate (CaCO3), is calcinated into calcium oxide (CaO), also known as quicklime. By adding water, the thus obtained calcium oxide can easily be transformed into calcium hydroxide [Ca(OH)2]. With the help of this highly exothermic reaction known as slaking, a soft and malleable paste called lime putty, is formed. Its hardening, which in its essence is the drying up of the material, continues as the chemical process of slow transformation of calcium hydroxide [Ca(OH)2]; the latter, when combined with carbon dioxide (CO2) from the air, reverts to calcium carbonate (CaCO3) and reaches substantial cohesion according to the reaction formula:
Ca (OH)2 + CO2 -» Ca CO3 + H2O
Dolomite (CaMg(CO3)2) disintegrates completely at temperatures between 940 °C and 1230 °C following the formula:
Ca Mg (CO3)2 -» CaO + MgO + 2CO2
Pure limestone is essential for obtaining finest-quality lime. Depending on its concentration, magnesium oxide (MgO) can have different effects: low concentration (dolomite limestones) produces “lean” lime; higher concentration, on the other hand, means “fatter” lime.
Lime produced from dolomite can have higher resistance compared to that made of limestone in the short run; dolomitic lime could pose serious problems over time, because of its interaction with the atmosphere, causing the formation of magnesium sulfate crystals on the surface that can cause it to deteriorate.
Download
 |
Scarica l'intero contenuto del pannello informativo n.5 (formato pdf) |